- All Products
- CellAlign Myo - Myoblast alignement and differentiation

CellAlign Myo - Myoblast alignement and differentiation
https://www.idylle-labs.com/shop/cellalign-myo-myoblast-alignement-and-differentiation-971 https://www.idylle-labs.com/web/image/product.template/971/image_1920?unique=639b047A ready-to-use substrate that guide myoblast alignment and support differentiation into long multinucleated myotubes.
A technology developed by Fouzia Boulmedais and Muhammad Haseeb Iqbal (Institut Charles Sadron, Strasbourg, France)
Designed to align. Built to differentiate.
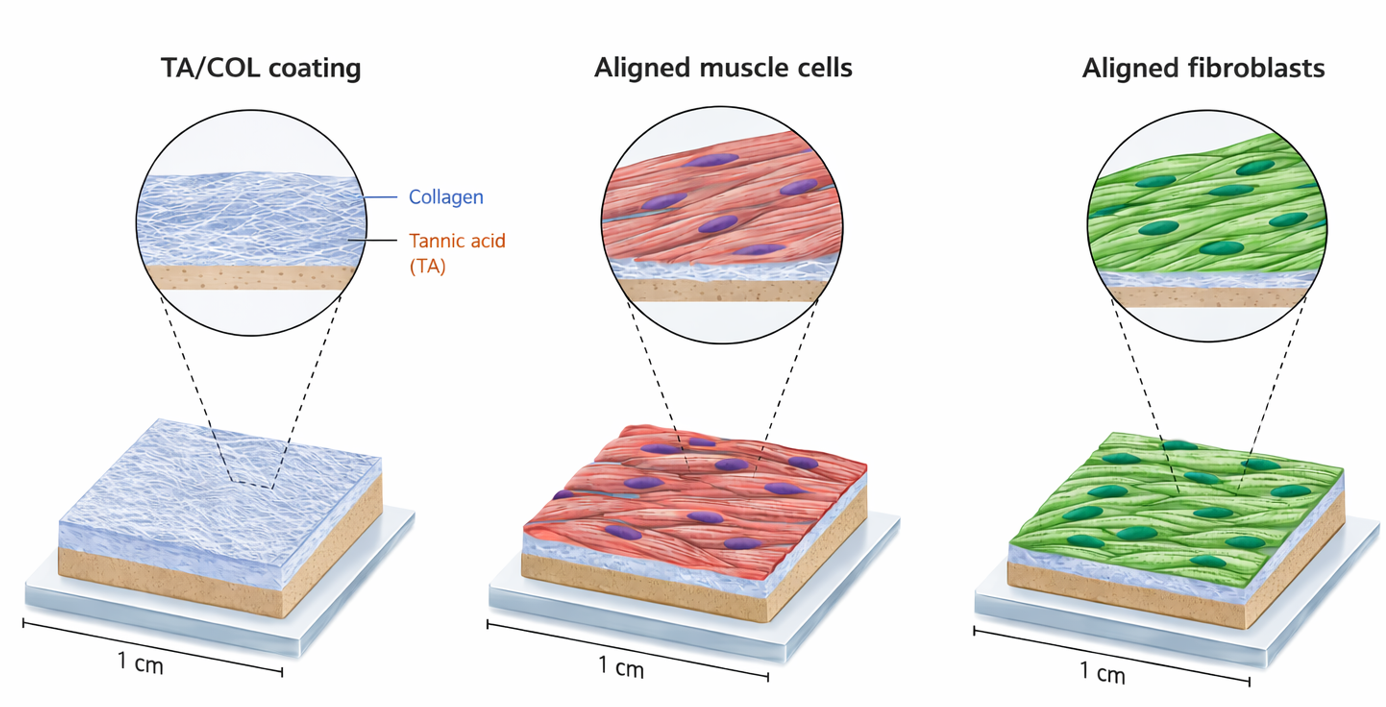
CellAlign is a pre-coated culture substrate based on oriented tannic acid / collagen films. It is designed for researchers who want a simple way to culture muscle cells on an aligned biomimetic surface. The oriented substrates guide myoblast alignment and support differentiation into long multinucleated myotubes.
CellAlign supports both structural organization and phenotypic maturation in muscle cell culture
Cellular alignment
CellAlign provides an oriented collagen-based surface that promotes the organization of myoblasts and fibroblasts and supports the formation of long, aligned myotubes.
Ready-to-use substrate
CellAlign is delivered as a finished coated substrate. Users receive a ready-to-use aligned surface, removing the need for in-house coating, patterning, or surface-preparation development.
Biocompatible
CellAlign combines collagen topographical guidance with tannic-acid-based film chemistry. Tannic acid release was linked to differentiation support, and showed local antibacterial activity.
Applications
-
Muscle cell culture
- Fibroblast cell culture
- Myoblast alignment studies
- Myotube formation assays
- Fibroblast alignment studies
- Cell morphology and focal adhesion studies
- In vitro models requiring anisotropic cell organization
* CellAlign substrates were also evaluated with fibroblasts, which adhered, spread, proliferated, and aligned on the oriented surface.
How does it work?
CellAlign works by combining directional surface guidance with bioactive TA/COL chemistry.
- The oriented collagen architecture gives cells a clear physical cue, helping them align and organize along the substrate.
- The tannic acid/collagen film chemistry supports the biological performance of the surface and contributes to the differentiation response observed in our muscle-cell model.
This combination helps create a more structured and biologically relevant culture environment than a conventional flat surface.
Validation highlights
- CellAlign Myo showed preserved nanostructure after 3 days in DMEM.
- Human myoblasts cultured on the oriented substrates formed long aligned myotubes after 12 days of differentiation.
- Tannic acid containing films showed local antibacterial activity and remained largely stable in physiological buffer.
Storage and shelf life
- Store at 4°C and protected from light
- For optimal performance, CellAlign should be used within 4 to 6 weeks of receipt
Discover the full protocol here
Download protocol
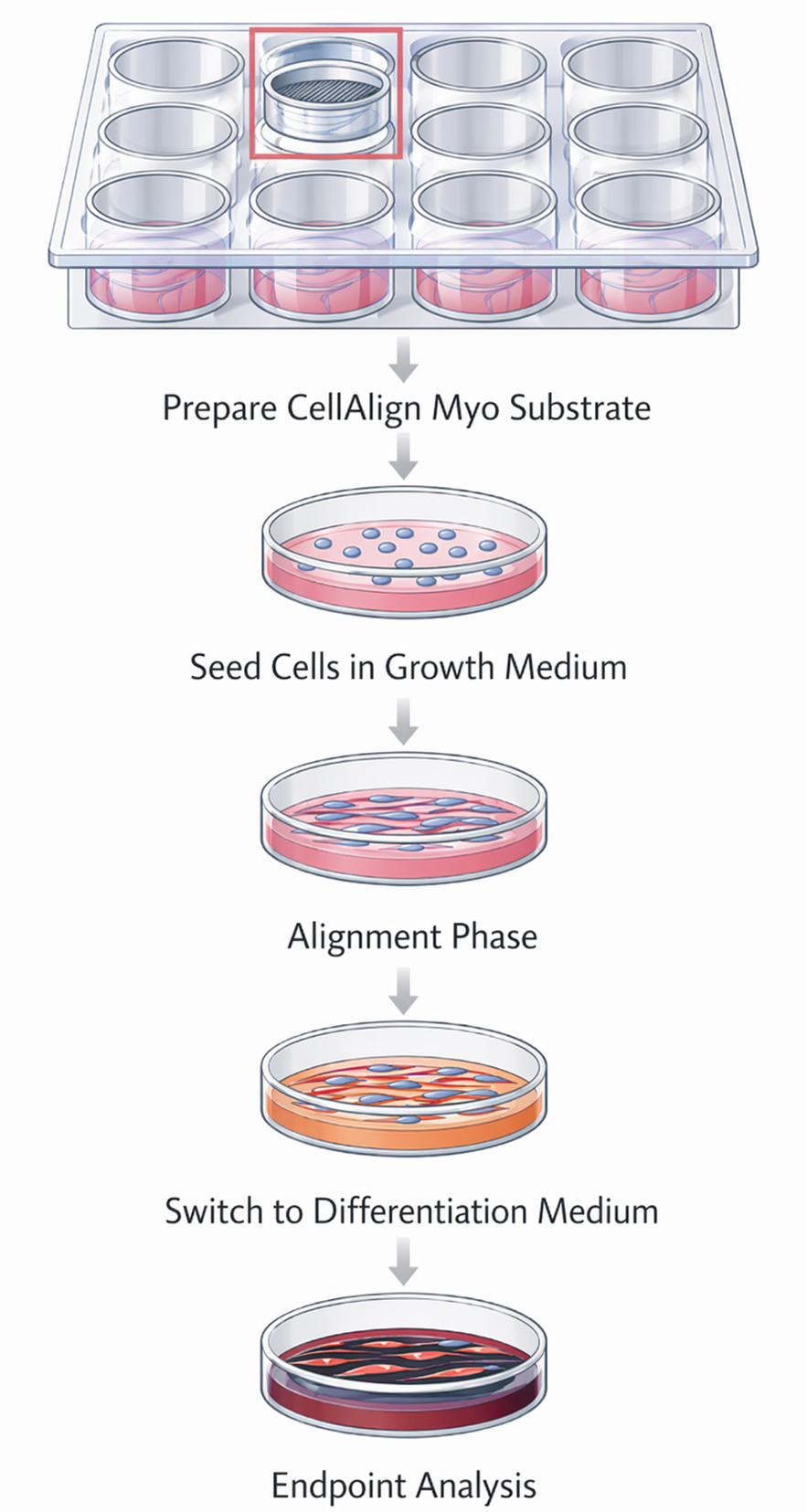
Protocol overview that shows the simplicity in adopting CellAlign substrates into your workflow:
Long, aligned human myotube formation on CellAlign Myo substrates
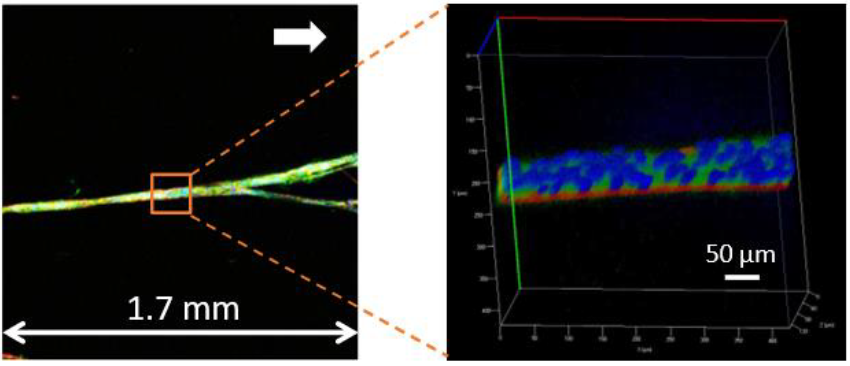
Confocal images of a long human myotube formed after 12 days of culture on CellAlign Myo substrates. The myotube extends over approximately 1.7 mm, with nuclei shown in blue, myosin heavy chain in green, and actin filaments in red. A 3D reconstructed zoom of the selected region is shown on the right.
Iqbal et al. 2022
CellAlign drives aligned myotube formation
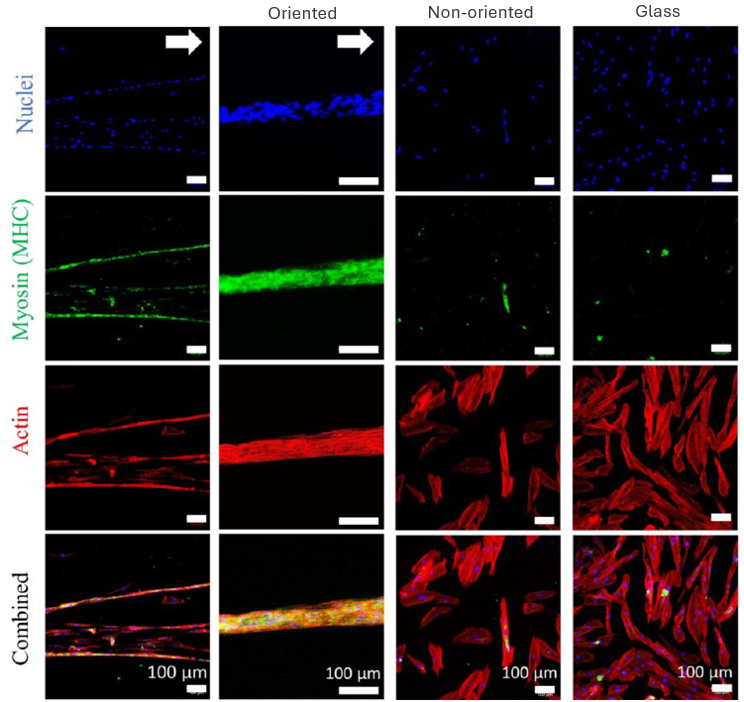
Immunostaining of human myoblast cultures highlights the biological effect of CellAlign: nuclei (blue), myosin heavy chain/MyHC (green), and actin (red) reveal the formation of long, aligned MyHC-positive myotubes on oriented substrates, compared with less organized differentiation on non-oriented substrates and glass. The white arrow marks the orientation direction of the substrate.
Iqbal et al. 2022
Myotubes elongate and align along the CellAlign orientation axis
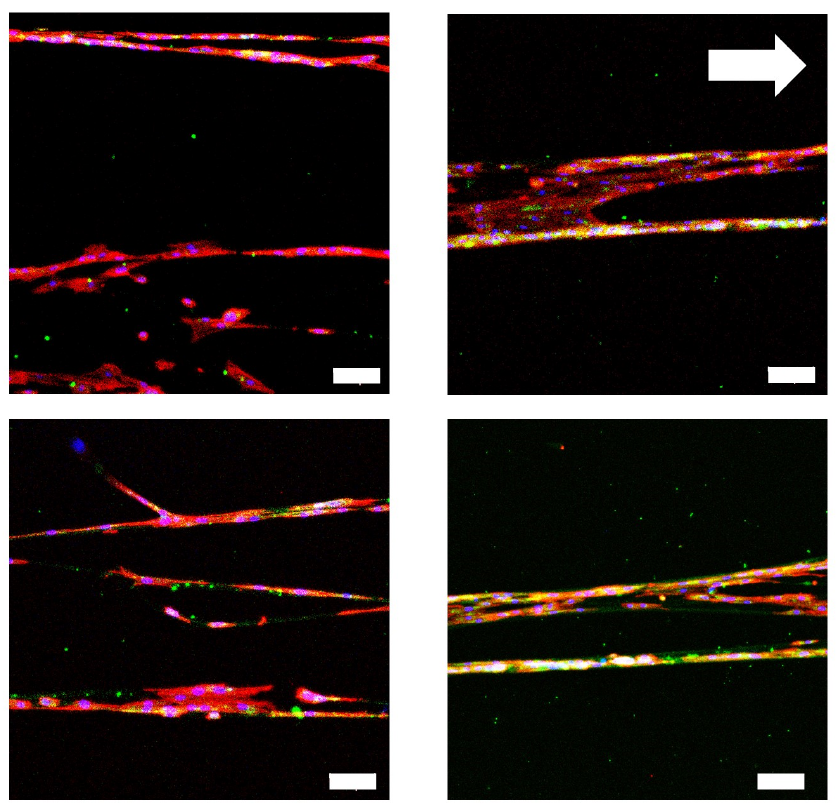
Human myoblasts form elongated, aligned MyHC-positive myotubes that follow the substrate orientation direction marked by the white arrow. Nuclei are shown in blue, myosin heavy chain (MyHC), a marker of muscle differentiation, is shown in green, and actin is shown in red.
Iqbal et al. 2022
CellAlign drives larger, longer, and more mature myotubes
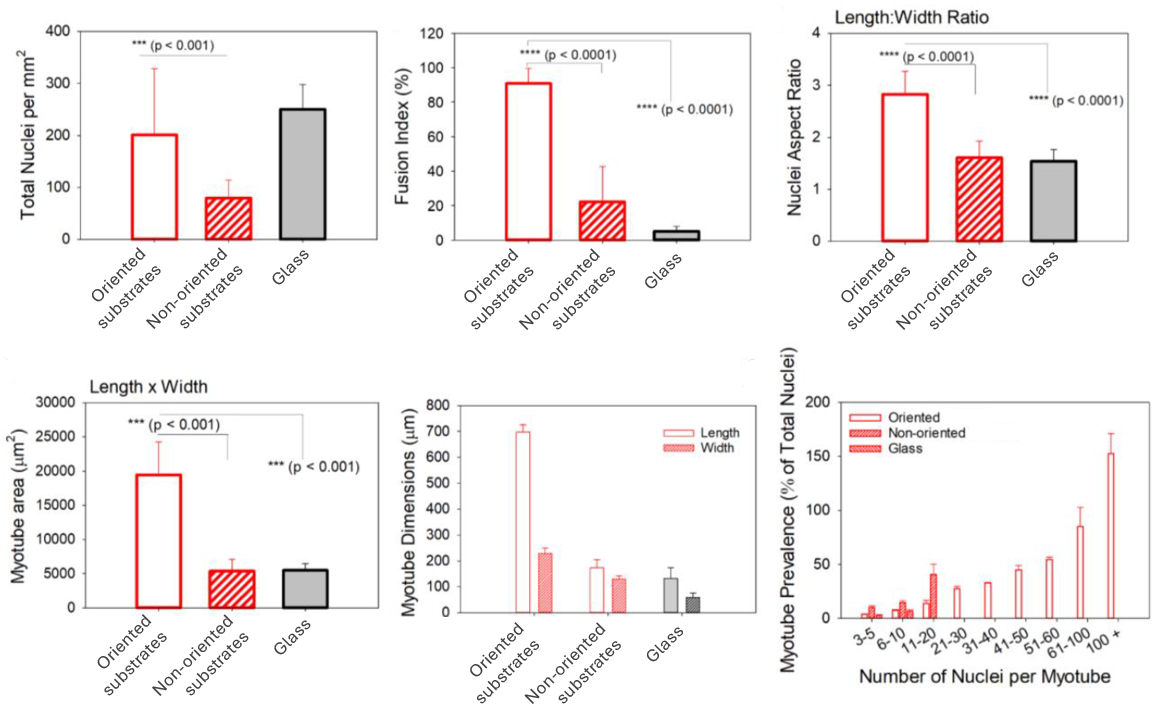
Quantitative analysis confirms the biological advantage of oriented substrates over non-oriented ones and glass controls. Compared with the other surfaces, the oriented substrates produce a much higher fusion index, more elongated nuclei, and markedly larger myotube area. They also generate longer myotubes and a much greater prevalence of highly multinucleated myotubes, indicating more advanced and organized muscle differentiation overall.
Iqbal et al. 2022
CellAlign promotes aligned and elongated cell organization
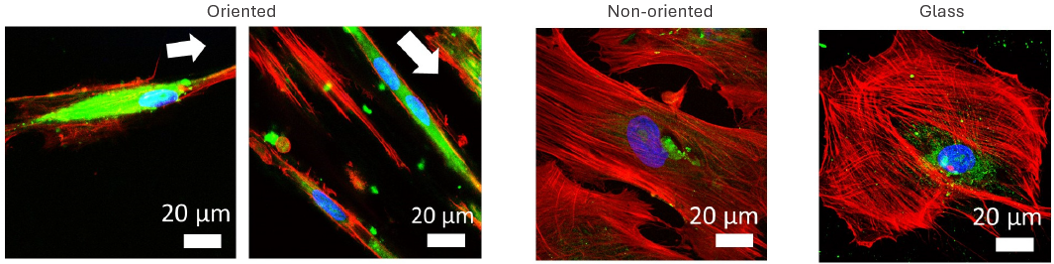
Confocal microscopy at higher magnification shows that CellAlign promotes a more elongated and directionally organized cell morphology. Nuclei are shown in blue, MyHC in green, and actin in red. The white arrows mark the substrate orientation direction, along which cells on the oriented TA/COL films become visibly more aligned than on non-oriented films or glass.
Iqbal et al. 2022
Fibroblasts cultured on CellAlign substrates showed elongated morphology and alignment along the oriented surface
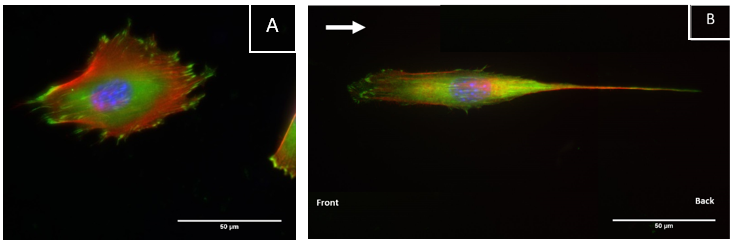
Nuclei are shown in blue, actin is shown in red, and vinculin, a focal adhesion marker, is shown in green.
Credits to: Emeline Pradel, Fouzia Boulmedais and Muhammad Haseeb Iqbal
CellAlign Myo promotes aligned cell organization
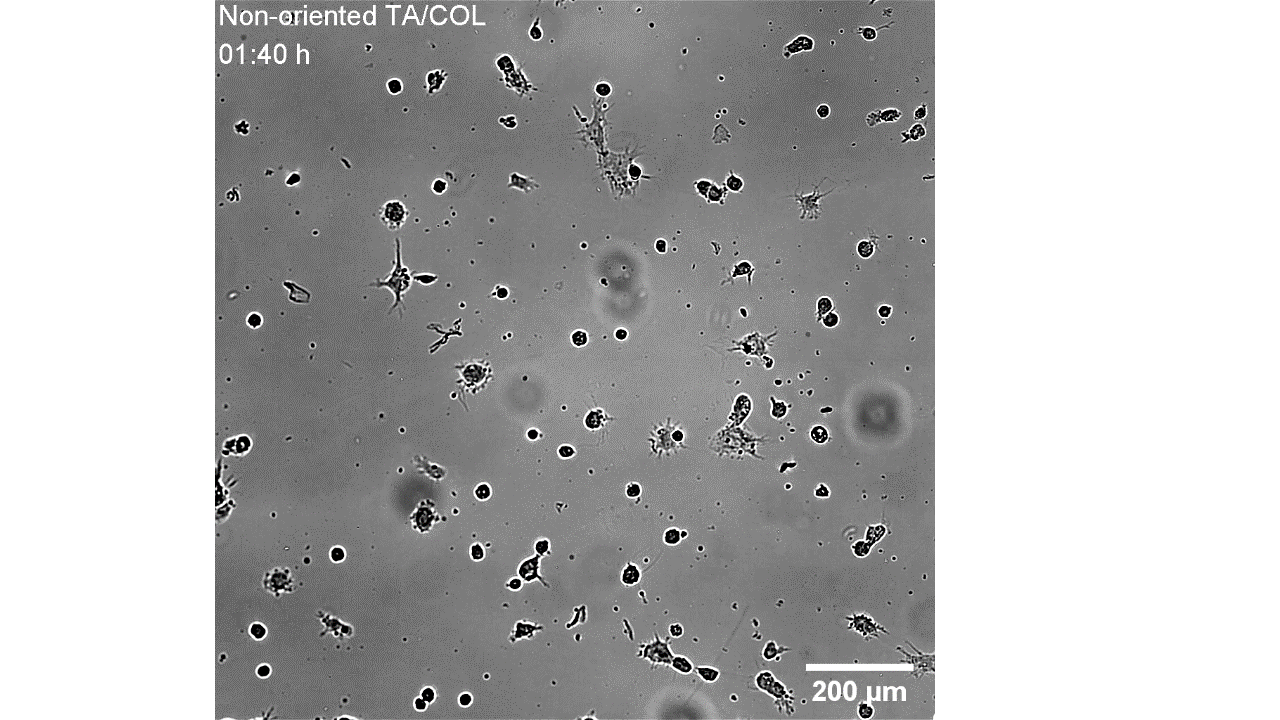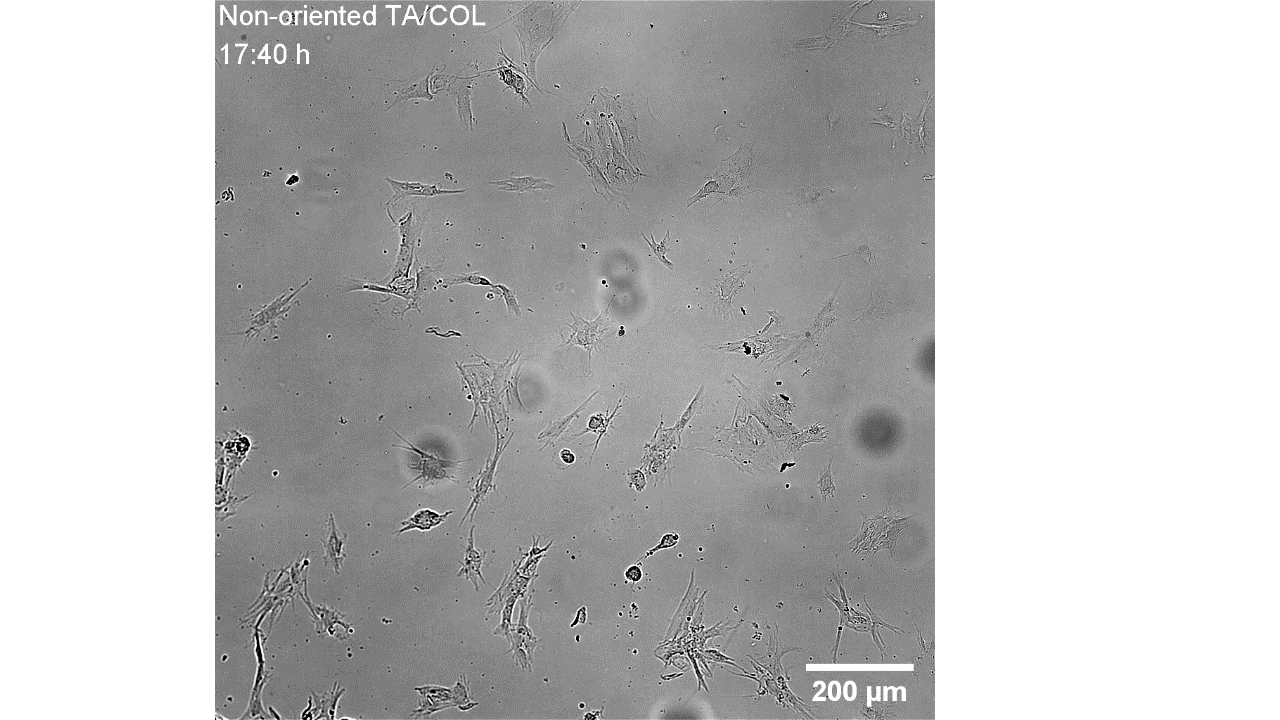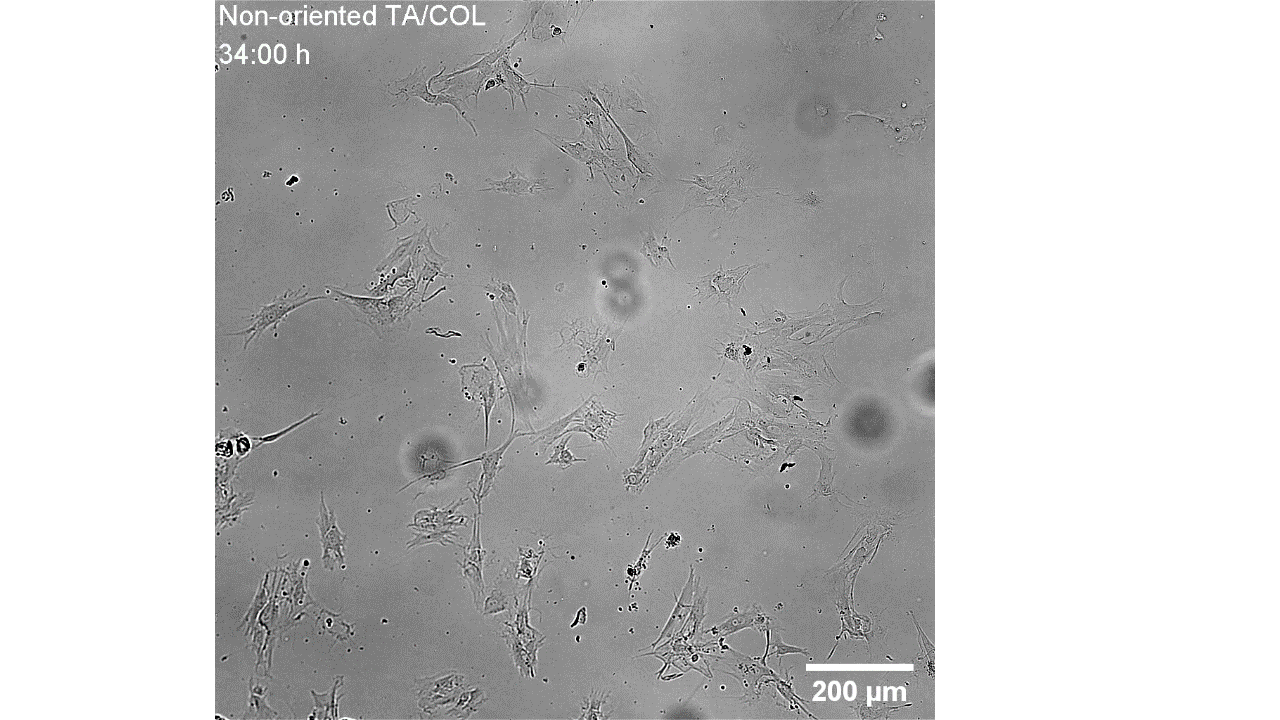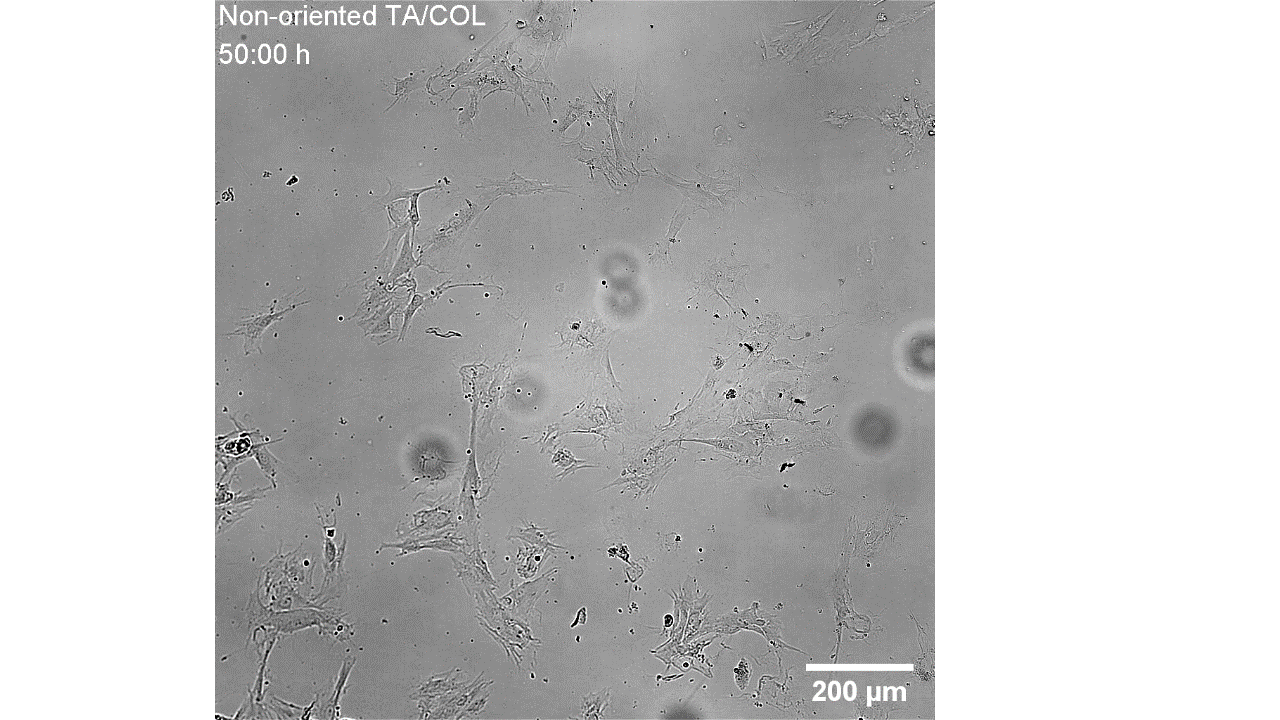
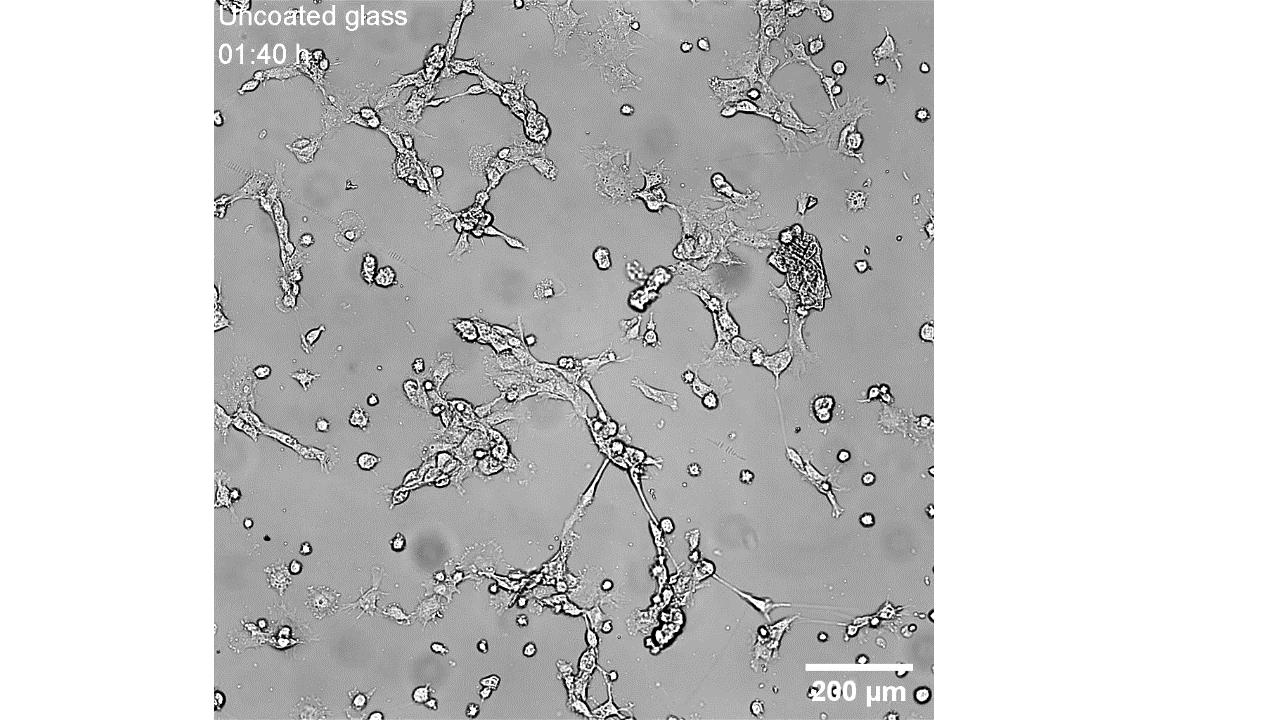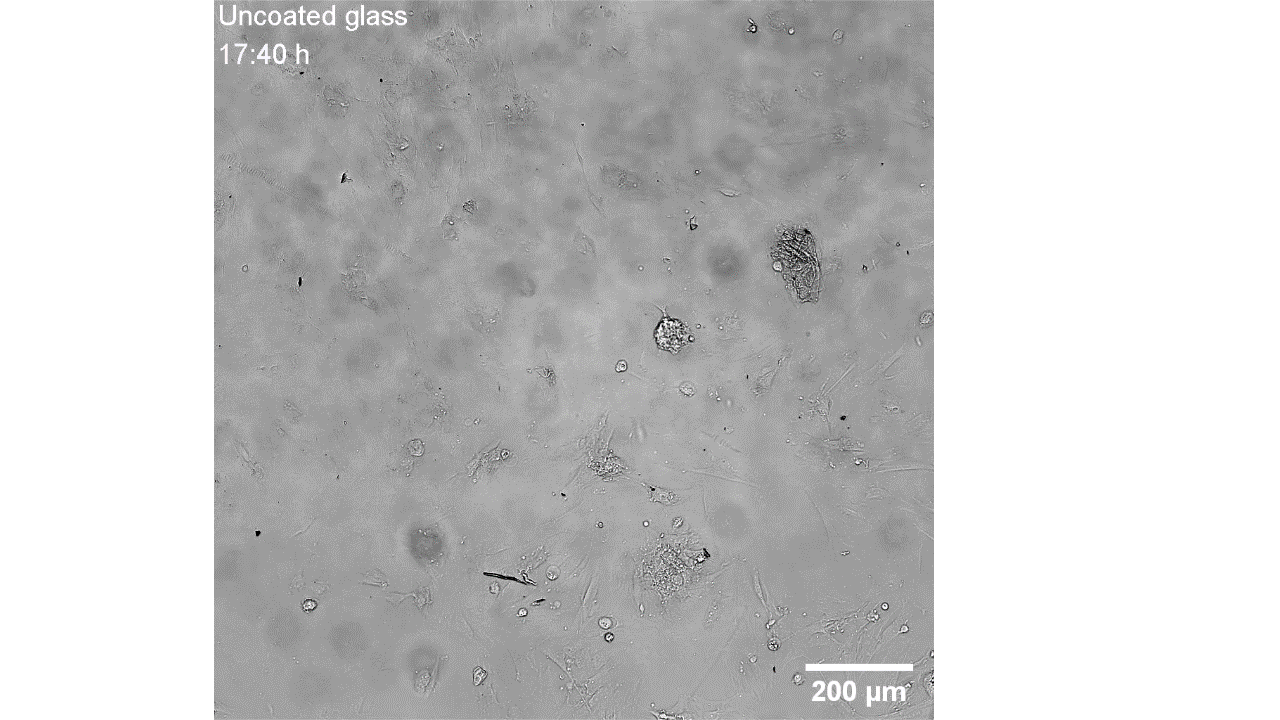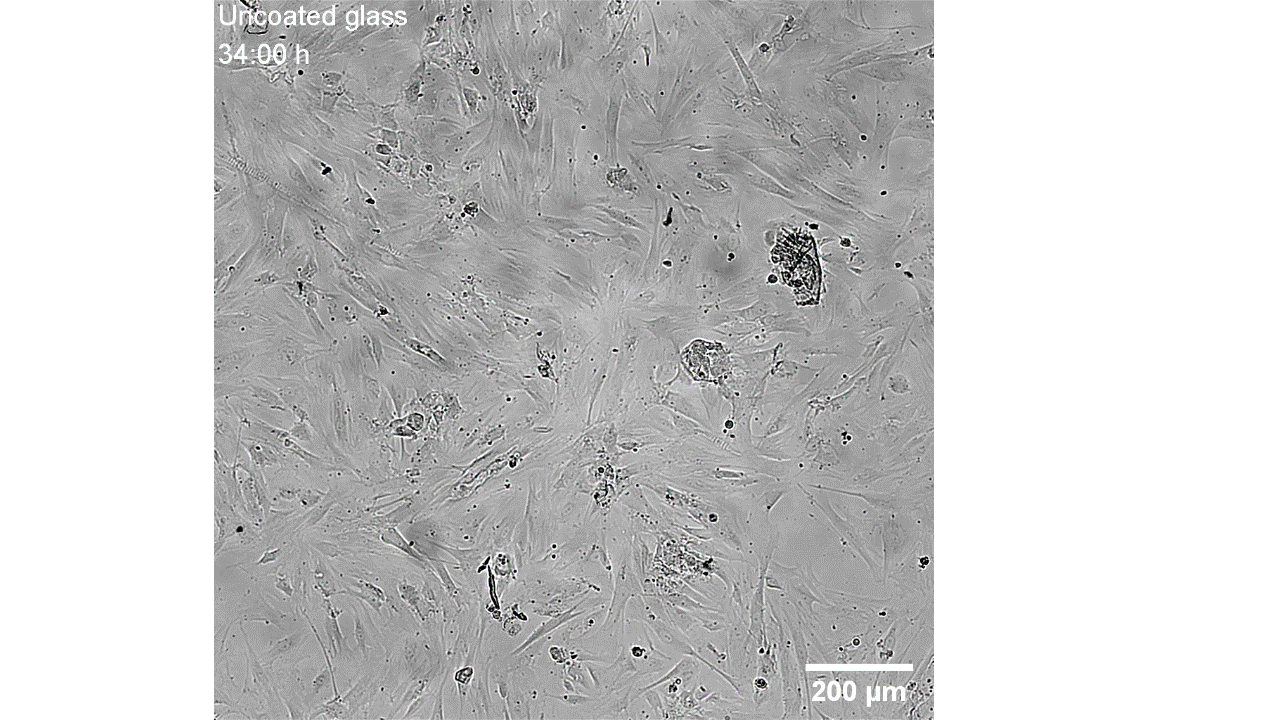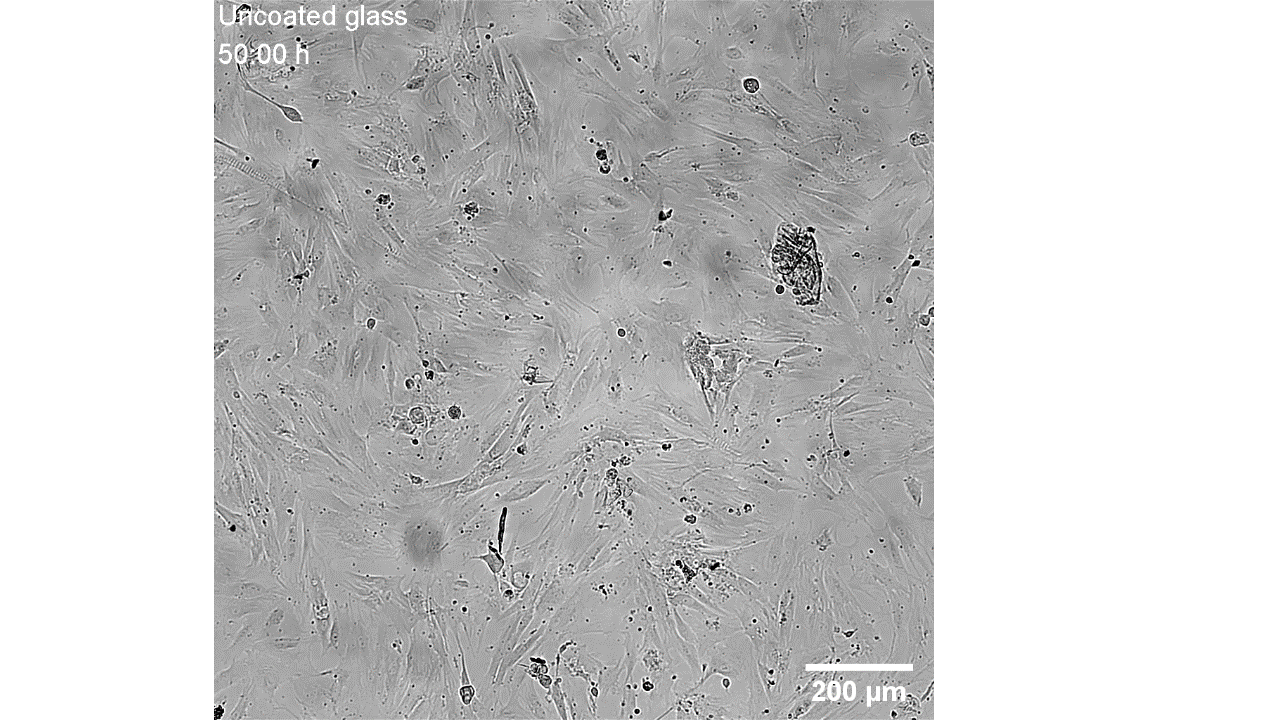
During the first 48 hours, oriented TA/COL films guided myoblasts to align and cluster, which likely supported later fusion and differentiation. On non-oriented films and uncoated glass, cell movement stayed random.
Iqbal et al. 2022
Unlike a flat standard surface, CellAlign provides an oriented micro/nanostructured collagen coating that helps guide cell organization. In the underlying data, this translated into more elongated, aligned, and mature myotube formation compared with non-oriented substrates and glass controls. By delivering this effect on a ready-to-use substrate, CellAlign can also reduce reliance on more complex, fabrication-intensive alignment methods such as micropatterning or lithography-based approaches.
So far, CellAlign has been validated mainly with myoblasts, which showed strong adhesion, alignment, and differentiation into long aligned myotubes on the oriented substrates. Fibroblasts have also been used as part of the substrate evaluation workflow, helping confirm that the oriented film architecture can be maintained under culture conditions. At present, muscle-cell culture remains the most strongly supported application, while additional studies are ongoing to extend the use of CellAlign to other cell types.
CellAlign is designed to act through both surface structure and bioactive chemistry. Its oriented collagen architecture gives cells a clear directional cue, encouraging aligned growth and organization. At the same time, tannic acid within the coating contributes to the biological performance of the substrate and supports myoblast differentiation. This combination led to the formation of long, aligned MyHC-positive myotubes with higher fusion and improved myotube morphology compared with non-oriented controls and glass.
Absolutely. CellAlign is specifically designed to create a more structured and biologically relevant culture environment for muscle-cell studies. Its oriented architecture helps guide myoblast alignment and supports the formation of elongated, organized myotubes, making it well suited for applications where muscle organization and directional cell growth are key readouts. Compared with non-oriented substrates and standard surfaces, the present results showed improved alignment and more mature myotube formation on the oriented films.
That is exactly its intended value. CellAlign is designed to provide a more structured culture environment for studies where cell orientation and organized myotube formation matter.
In CellAlign, tannic acid is incorporated into the coating in a way that remains compatible with myoblast culture. The data show that myoblasts on the oriented substrate successfully adhere, align, and differentiate into long myotubes. Data also support that while high concentrations of tannic acid in solution can reduce viability, the lower levels relevant to these substrates conditions remain compatible with the cells.
Brush-Induced Orientation of Collagen Fibers in Layer-by-Layer Nanofilms: A Simple Method for the Development of Human Muscle Fibers.
Iqbal MH, Revana FJR, Pradel E, Gribova V, Mamchaoui K, Coirault C, Meyer F, Boulmedais F. ACS Nano. 2022 Dec 27;16(12):20034-20043.
doi: 10.1021/acsnano.2c06329. Epub 2022 Oct 27. PMID: 36301714.
Effect of the Buffer on the Buildup and Stability of Tannic Acid/Collagen Multilayer Films Applied as Antibacterial Coatings
Muhammad Haseeb Iqbal, André Schroder, Halima Kerdjoudj, Christian Njel, Bernard Senger, Vincent Ball, Florent Meyer, and Fouzia Boulmedais
ACS Applied Materials & Interfaces 2020 12 (20), 22601-22612
DOI: 10.1021/acsami.0c04475
Everspark technology has been intially developed by Karine Monier, Arnaud Favier and Christophe Place and and published in Scientific Reports:
Provost, A., Rousset, C., Bourdon, L. et al. Innovative particle standards and long-lived imaging for 2D and 3D dSTORM. Sci Rep 9, 17967 (2019). https://doi.org/10.1038/s41598-019-53528-0
Publications:
Nanoscale engagement and clusterization of Programmed death ligand 1 (PD-L1) in the membrane lipid rafts of Non-Small Cell Lung Cancer cells
Martina Ruglioni, Simone Civita, Tiziano Salvadori, Sofia Cristiani, Vittoria Carnicelli, Serena Barachini, Iacopo Petrini, Irene Nepita, Marco Castello, Alberto Diaspro, Paolo Bianchini, Barbara Storti, Ranieri Bizzarri, Stefano Fogli and Romano Danesi bioRxiv 2022.08.09.503318; doi: https://doi.org/10.1101/2022.08.09.503318
HIV-1 diverts actin debranching mechanisms for particle assembly and release in CD4 T lymphocytes
Rayane Dibsy, Erwan Bremaud, Johnson Mak, Cyril Favard, Delphine Muriaux
BioRxiv, December 16, 2022. doi. 10.1101/2022.12.15.520580
Fluorescent Polymer-AS1411-Aptamer Probe for dSTORM Super-Resolution Imaging of Endogenous Nucleolin
Fabre L, Rousset C, Monier K, Da Cruz-Boisson F, Bouvet P, Charreyre MT, Delair T, Fleury E, Favier A. Biomacromolecules. 2022 May 12. doi: 10.1021/acs.biomac.1c01706. PMID: 35549176
Comparative analysis of ChAdOx1 nCoV-19 and Ad26.COV2.S SARS-CoV-2 vector vaccines.
Michalik S, Siegerist F, Palankar R, Franzke K, Schindler M, Reder A, Seifert U, Cammann C, Wesche J, Steil L, Hentschker C, Gesell-Salazar M, Reisinger E, Beer M, Endlich N, Greinacher A, Völker U. Haematologica. 2022 Apr 1;107(4):947-957. doi: 10.3324/haematol.2021.280154. PMID: 35045692
Metabolic biorthogonal labeling and dSTORM imaging of peptidoglycan synthesis in Streptococcus pneumoniae
Jennyfer Trouve, Oleksandr Glushonkov and Cecile Morlot
Star Protocols, December 13, 2021. doi: 10.1016/j.xpro.2021.101006
Insights in ChAdOx1 nCoV-19 vaccine-induced immune thrombotic thrombocytopenia.
Greinacher A, Selleng K, Palankar R, Wesche J, Handtke S, Wolff M, Aurich K, Lalk M, Methling K, Völker U, Hentschker C, Michalik S, Steil L, Reder A, Schönborn L, Beer M, Franzke K, Büttner A, Fehse B, Stavrou EX, Rangaswamy C, Mailer RK, Englert H, Frye M, Thiele T, Kochanek S, Krutzke L, Siegerist F, Endlich N, Warkentin TE, Renné T.
Blood. 2021 Dec 2;138(22):2256-2268. doi: 10.1182/blood.2021013231. PMID: 34587242
Superresolution Microscopy of Drosophila Indirect Flight Muscle Sarcomeres.
Szikora S, Novák T, Gajdos T, Erdélyi M, Mihály J. Bio Protoc. 2020 Jun 20;10(12):e3654. doi: 10.21769/BioProtoc.3654. eCollection 2020 Jun 20. PMID: 33659324