Amandine Virlogeux
Eve Moutaux
Wilhelm Christaller
Aurélie Genoux
Julie Bruyère
Elodie Fino
Benoit Charlot
Maxime Cazorla
Frédéric Saudou
Scientists recreate in vitro a physiological neuronal network by implementing a microfluidics device to control the neuronal connectivity and function. This tool allows not only the modelling of pathological networks, as found in Huntington’s disease, but also the cellular compartmentalization to decipher the molecular mechanisms responsible for the pathology and the drug screening to rescue the impacted biological processes.
Publication: Reconstituting Corticostriatal Network on-a-Chip Reveals the Contribution of the Presynaptic Compartment to Huntington’s Disease
#Microfluidics #Axonal&DendriticTransport #Mitochondria #Glutamate #BDNF #Synapse

A blog post by Hélène Vitet
I have a PhD in neuroscience with an engineering background, this might be why I am interested in combining biology and technology. During my PhD, I have studied intracellular dynamics in neurons using microfluidics and I am currently pursuing a postdoc focusing on circadian rhythm establishment in the brain. Writing on the Idylle blog is more than just a way to keep me updated about new innovative tools, it’s about sharing and promoting state-of-the-art techniques that deserve to get your attention.
.png)
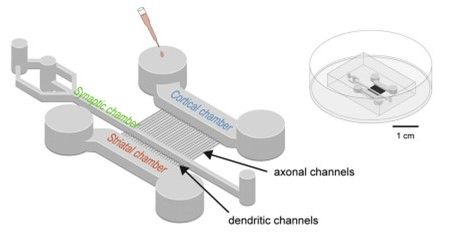
Figure 1: Scheme of the microfluidics device containing 3 chambers to recreate a neuronal network. From Virlogeux et al., 20
Why should you use this microfluidic device?
Neurons are ultra-polarized cells, it is thus crucial to use tools that discriminate subcellular compartments. You want to know if you are looking at a dendrite or an axon and if it is connected or mature enough. This is especially true when you want to monitor dynamics; when you spot a moving vesicle you might want to know where it goes, in which direction.
1. Time control
You can control the maturation of your network, which directly impact its connectivity and consequently its function. You can choose to study axonal growth, postsynaptic signaling, or both!
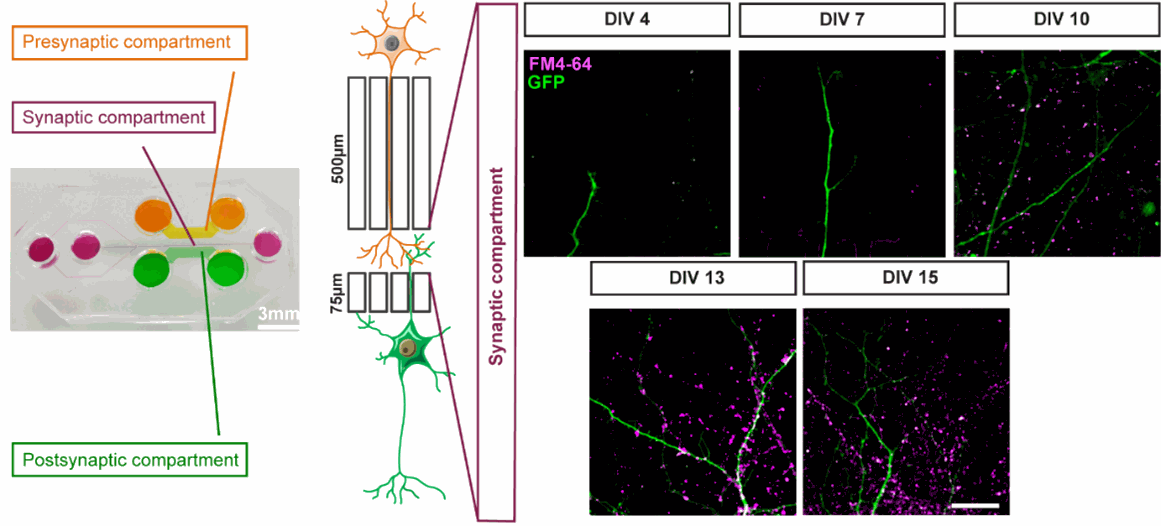
Figure 2: Time control of the neuronal network connectivity. Functional synapses were detected using FM4-64 dye (purple) that labels active presynaptic boutons on GFP dendrites (green) upon 50 mM KCl stimulation. Modified from Bruyere et al., 2020
2. Space control
You can control the organization of your network. This feature allows you to manipulate directly and specifically the presynaptic compartment (axons), the synaptic compartment (synapses) or the postsynaptic compartment (dendrites).
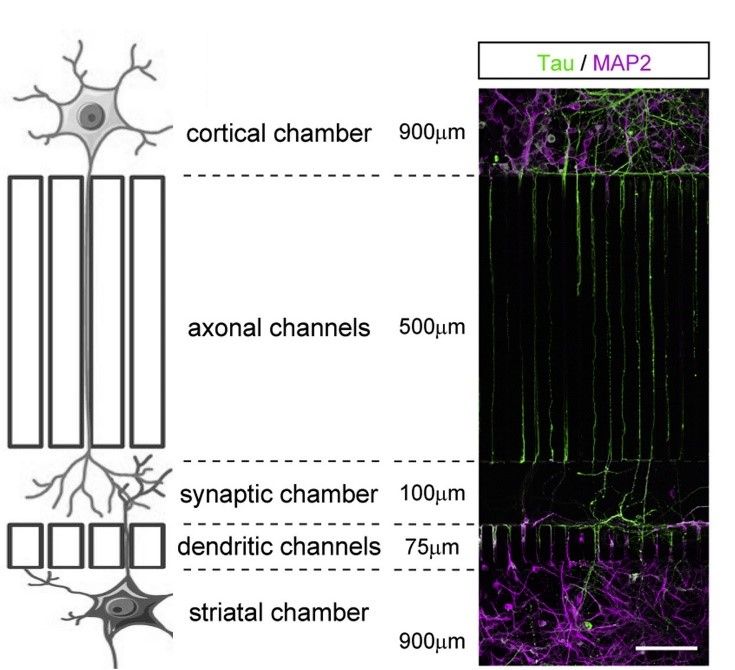
Figure 3: spatial control of the neuronal network. Tau is used as an axonal marker and MAP2 is used as a dendritic marker. Modified from Bruyere et al., 2020 and Virlogeux et al., 2018
3. Study many biological processes
From protein levels to network function or subcellular dynamics, this device can be the basis of many of your experiment’s setups.
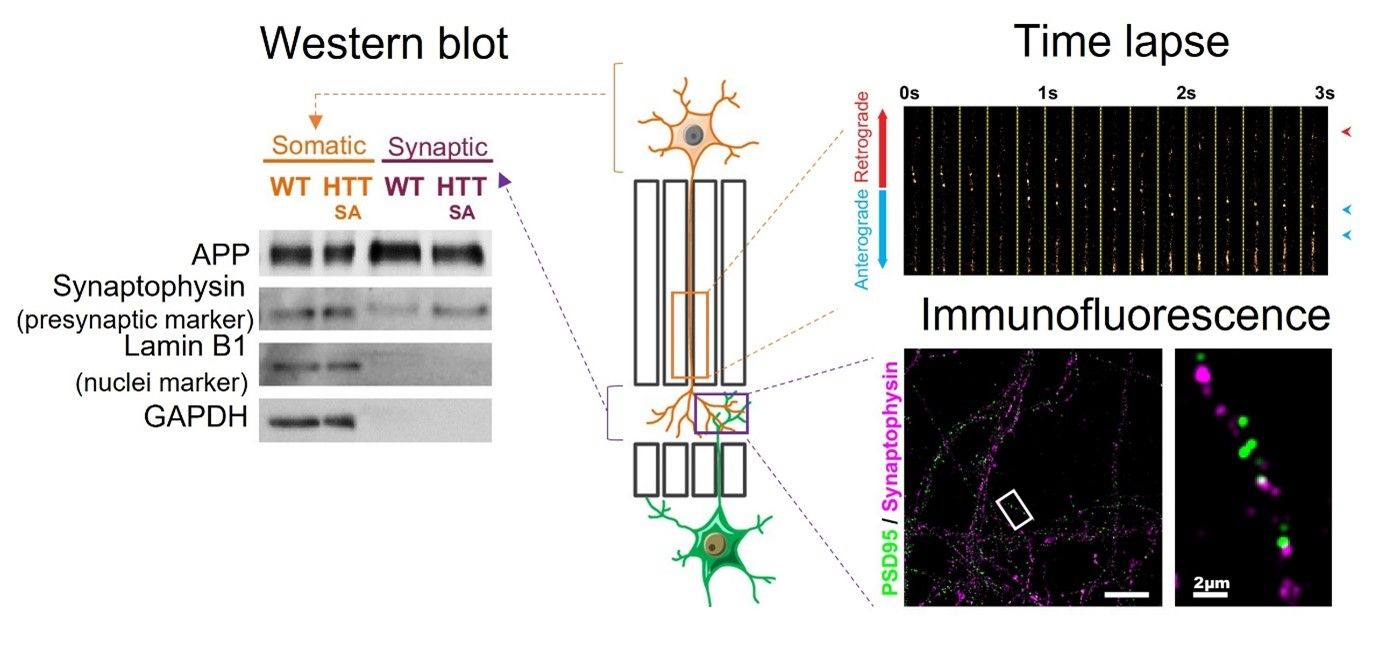
Figure 4: common biochemistry and biocellular experiments can be performed with this microfluidics system. Modified from Bruyère et al., 2020
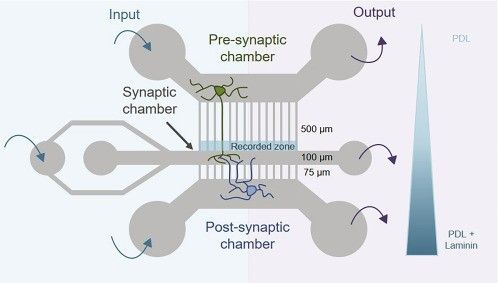
Figure 5: spatial control of the neuronal network trough physical and chemical neurite properties.
From Lenoir et al., 2021
How does this microfluidic device work?
What make the difference between this microfluidic device and the one it originates from (Taylor et al.,2010) are both its design and the chemicals applied. Indeed, the axons in the presynaptic compartment are discriminated from the dendrites by their length; the axon channels are longer. Then, the laminin gradient limits the number of axons from the postsynaptic chamber to reach the synaptic chamber.
What can I study with this microfluidic device?
When your network has reached the desired maturation state, you just need to play! Add lentiviruses when and where you want and then, study subcellular dynamics (vesicular transport, mitochondrial behavior), network function (neurotransmitter release, postsynaptic transmission, connectivity, synchronization) or protein (co)localization. You can also make western blots out of the neurons you plated to assess the protein level in a compartment. Finally, the beauty of this tool is that it can be used for any other neuron type or any other neurodegenerative disease model.
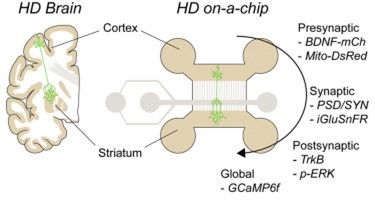
Figure 6: Tools to study the neuronal network function. From Virlogeux et al.,2018
In a nutshell
This new microfluidics device is interesting in the sense that you can organize and control the neurons you would have grown freely in a petri dish. Moreover, this in vitro reconstitution of a neuronal network recapitulates a brain connection and may thus limit the animal use in the future, for drug screening for example.
References:
Bruyère, J., Abada, Y.-S., Vitet, H., Fontaine, G., Deloulme, J.-C., Cès, A., Denarier, E., Pernet-Gallay, K., Andrieux, A., Humbert, S., Potier, M.-C., Delatour, B., & Saudou, F. (2020). Presynaptic APP levels and synaptic homeostasis are regulated by Akt phosphorylation of huntingtin. ELife, 9. https://doi.org/10.7554/eLife.56371
Virlogeux, A., Moutaux, E., Christaller, W., Charlot, B., & Bruye, J. (2018). Reconstituting Corticostriatal Network on-a-Chip Reveals the Contribution of the Presynaptic Compartment to Huntington ’ s Disease Article Reconstituting Corticostriatal Network on-a-Chip Reveals the Contribution of the Presynaptic Compartment to Huntingt . 110–122. https://doi.org/10.1016/j.celrep.2017.12.013
Lenoir, S., Genoux, A., Agasse, F., Saudou, F., & Humbert, S. (2021). Recreating mouse cortico-hippocampal neuronal circuit in microfluidic devices to study BDNF axonal transport upon glucocorticoid treatment. STAR Protocols, 2(1). https://doi.org/10.1016/j.xpro.2021.100382
Taylor, A. M., Dieterich, D. C., Ito, H. T., Kim, S. A., & Schuman, E. M. (2010). Microfluidic Local Perfusion Chambers for the Visualization and Manipulation of Synapses. Neuron, 66(1), 57–68. https://doi.org/10.1016/j.neuron.2010.03.022
MORE LIFE SCIENCE TOOLS TO FOLLOW
You want the latest on life science tools to follow, test and more?